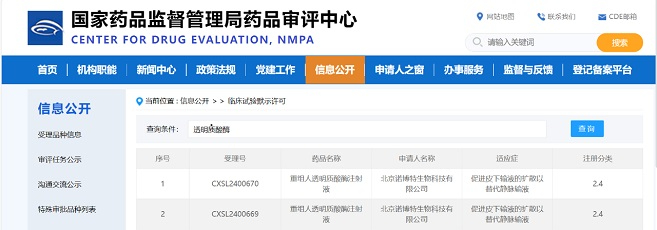
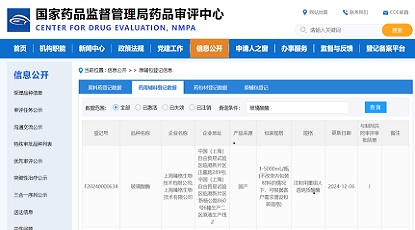
Congratulations to Latticon Antibody Therapeutics, Inc.("Latticon") on obtaining the Investigational New Drug (IND) approval from the Food and Drug Administration (FDA) for its globally pioneering "α-clash" IL-2 antibody fusion protein drug, LAT010.
As the antibody CMC service provider for Latticon, Zencore Biologics is honored to offer comprehensive CMC support from Sequence to IND submission, aiding Latticon in obtaining IND approval for its new drug project.
Zencore Biologics sincerely wish that Latticon will make smooth clinical progress and be launched as soon as possible, which will bring the hope of cure to patients. Zencore Biologics will always adhere to providing high-quality, efficient and comprehensive CDMO services for global partners.